Home page
Welcome to the Male Infertility and Germ Cell Biology laboratory, we are located in the Bio 21 Institute and are part of the School of BioSciences at The University of Melbourne

Male infertility and germ cell biology
We aim to identify key mechanisms required for male germ cell development, the aetiology of human male infertility and the interplay between fertility and health. This is achieved using a range of genomic, biochemical and cell biological methods, including the development of unique model systems.
Research overview
Male fertility research provides data of three-fold value:
- Male infertility is a major medical problem affecting 1 in 20 Australian men of reproductive age. For the majority of these men the underlying aetiology is unknown and thus, targeted therapies cannot be applied
- An enhanced understanding of the mechanisms of spermatogenesis (the process of sperm production) may provide opportunities for contraceptive intervention;
- Spermatogenesis has proven to be an extremely productive system within which to discover molecules and processes of fundamental importance to cell biology and human health.
Each of the fields of stem cells, cilia biology, epigenetics and DNA repair had their origins in the testis. In order to produce 1,000 sperm per heart beat, as humans do, stem cells must divide continuously, the DNA breaks inherent in cell division and in meiosis in particular must be faithfully and rapidly repaired, and the thousands of genes involved in forming the highly condensed, and motile sperm must be tightly regulated at each of a transcriptional, translational and post-translational level. Unlike the majority of tissues wherein these processes occur only in discrete developmental processes or in response to challenge, within the testis they occur continuously and on a large scale, thus making spermatogenesis an outstanding system within which to discover novel pathways and to define their in vivo function.
Research focus
Cilia/flagellar development and function, genetic causes of human infertility, the importance of epigenetic regulation in male fertility, sperm head shaping and the transcriptional and translational control of germ cell-expressed genes.
Want to hear more? Click on the News tab above
Our available projects revolve around the major lab themes:
- The core processes of sperm construction and function
- Understanding the genetic causes of male infertility
CURRENT PROJECTS – get in contact now for 2025 Honours, Masters and PhD:
Title: Investigating the role of a novel gene in spermatogenesis and male fertility.
Location: Bio21 Institute
Supervisors: Dr. Brendan Houston, Prof. Moira O’Bryan
Contact: moira.obryan@unimelb.edu.au
Project type: Hons, MSc or PhD project
Background and Project Description
Male infertility is a common condition that affects at least 7% of men and is often caused by genetic factors. In line with the variety of cellular processes involved in sperm production, the testis expresses around 17,000 genes and the role of many of these is unknown. As part of a collaborative role in the International Male Infertility Genomics Consortium, we have identified genetic variants in several genes to be a likely cause of infertility in several men who do not produce sperm.
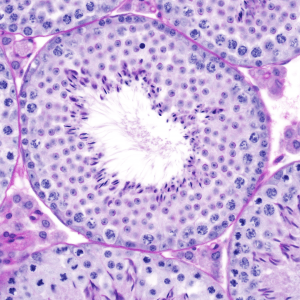
This project will involve fertility ‘phenotyping’ of knockout males using a standard pipeline and expert training available in the Male Infertility and Germ Cell Biology lab. You will assess whether knockout males are infertile and validate the knockout model. The student will also define the consequences of gene loss on the histology of the testis and male germ cells, daily sperm production and sperm function, if they are present. You will also define the localisation of the gene of interest in the wild type testis.
Title: Inter-species differences in sperm structure.
Location: Bio21 Institute
Supervisors: Dr Jessica Dunleavy, Dr. Brendan Houston, Prof. Moira O’Bryan
Contact: jessica.dunleavy@unimelb.edu.au
Project type: MSc project
Background and Project Description
Sperm structure varies greatly between species. The reason for, and the consequences of, these differences are poorly understood. Such differences occur in the sperm head, and the tail including in the mitochondrial sheath. In sperm, mitochondria are contained with the midpiece and separated from the remainder of the cell where they generate energy for sperm function, including for sperm tail motility.
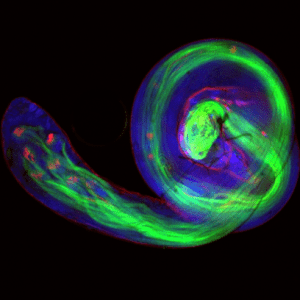
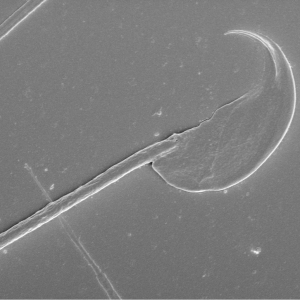
This project will involve an investigation of the sperm structure at a three-dimensional level. This will include an analysis of sperm head shape, the mitochondrial sheath structure and the sperm tail as a whole and comparisons across species (mammals, fish, insects, etc.) using scanning and transmission electron microscopy. Collectively, this will allow insights into species differences in sperm structure and function and may feed into our understanding of the consequences of differences in evolutionary pressures.
Other areas of interest:
Investigating the role of the cytoskeleton in spermatogenesis.
Email jessica.dunleavy@unimelb.edu.au for more info
Characterising novel conditional spermiogenesis drivers in Drosophila melanogaster
Email brendan.houston@unimelb.edu.au for more info
Prof. Moira O’Bryan
Lab head and Dean of Science at The University of Melbourne
Moira is a world leader in the fields of male fertility, male germ cell development and sperm function. She is an ARC college of experts panel member (2018-2020), has delivered numerous plenary presentations, has won multiple high-ranking awards, and has held continuous grants from the ARC and NHMRC for 26 years. Her lab has two largely overlapping themes:
- Understanding the mechanisms of sperm production and function, i.e. ‘how to build functional sperm’
- Defining the genetic causes of male infertility
Moira is a steering member of the International Male Infertility Genomics Consortium
Dr. Brendan Houston (he/him)
Postdoctoral Research Fellow
Brendan works on defining novel genetic causes of human male infertility using a variety of model systems (mice, flies and cultured cells). These models are based on high confidence genetic variants found in infertile men from the International Male Infertility Genomics Consortium and The Genetics of Male Infertility Initiative. He was awarded his PhD from The University of Newcastle, Australia (supervised by L/Prof. John Aitken, Prof. Brett Nixon, Dr. Geoffry De Iuliis and Dr. Bruce King) in 2018 and has worked in the O’Bryan lab since. Brendan is a past Rutherford fellow (UUKi) and is available as a PhD supervisor, so feel free to contact him for more info.
Dr. Jessica Dunleavy
Postdoctoral Research Fellow
Jess works on understanding the fundamental mechanisms that drive mammalian sperm production using multiple knockout mouse models. Her research mainly centres around cytoskeletal dynamics and their relevance to male fertility, with particular focus on the unique cytoskeletal structures, regulators and protein transport pathways that feature during male meiosis and spermatid remodeling. She was awarded her PhD from Monash University (supervised by Prof. Moira O’Bryan and Prof. John Carroll) in 2018. Prior to that, she worked under the supervision of Prof. Neil Gemmell and Dr Sheri Johnson at the University of Otago, studying the effect of paternal age on fertility in Zebrafish. Jess and Moira currently hold an NHMRC Ideas Grant investigating the mechanisms of mitochondrial loading onto the sperm tail during spermiogenesis and an ARC Discovery Project investigating the role of tubulin post-translational modifications in spermatogenesis. Jess is available as a PhD supervisor, so feel free to contact her for more info.
Anne O’Connor (Bsc(Hons))
Laboratory manager
Anne manages the lab and has significant experience in working with protein production and assessment, hormone assays, antibody development and various other techniques. Anne has worked with Moira for many years and has been involved in the characterisation of multiple knockout mouse models, including several mutagenesis studies.
Gemma Stathatos
Postdoctoral Research Fellow
Joseph Nguyen (BSc(Hons))
Research assistant
Joseph is a research assistant that previously investigated research questions into the biology and functionality of neural receptors, specifically in Drosophila. This has given him broad experiences into molecular techniques which include cloning, recombineering, construct design and assembly. He is also experienced in microscopy as well as protein tagging and visualisation.
PhD students
Maddison Graffeo
Maddison investigates the role of a novel coiled-coil containing protein in male fertility and sperm function. She is co-supervised by Dr. Brendan Houston and Dr. Jessica Dunleavy.
Visiting researchers
Hongyi Yang (2024-2025) – visiting from China (The First Affiliated Hospital of Zhengzhou University)
Hongyi is a visiting research and medical doctor. He is investigating the role of a coiled-coil protein in male fertility.
Gülizar Saritas (November 2023-April 2024) – visiting from Denmark (University of Copenhagen)
Güli is a PhD student in the Almstrup group (Copenhagen) and is training in mouse model phenotyping.
Zoé François (February – May 2024) – visiting from France (Claude Bernard University Lyon 1)
Zoé is a Masters student with a background in electron microscopy and is learning new techniques in molecular biology, focusing on models of abnormal sperm morphology.
Honours students – 2024
Jack Deane – Bachelor of Science Honours
Jack is investigating the role of osmoregulation in sperm function.
Research technicians
None at current
See all of Moira’s publications:
See all of Jess’s publications:
Prof. Moira O’Bryan – Dean of Science
Old Geology Building (Building 155)
The University of Melbourne
Royal Parade
Victoria 3010
email: moira.obryan@unimelb.edu.au
Executive Assistant – Helen Godsall
email:
phone: +61390353011
2024
- Brendan and Moira contributed to a study in Molecular Cell “C19ORF84 connects piRNA and DNA methylation machineries to defend the mammalian germline”.
- Mia Everrett joined our lab as a casual research technician.
- Hongyi Yang and Zoe Francois have joined us for research projects; Jack Deane has joined us for his honours year.
2023
- Sam Cheers passed his PhD and is now working in Cambridge in the Reid Lab.
- Moira led and Brendan contributed to an important expert recommendation paper “Frequency, morbidity and equity—the case for increased research on male fertility” published in Nature Review Urology. This was the results of a combined effort of 26 researchers globally.
- Jess and the lab led a study published in Development entitled “Katanin A-subunits KATNA1 and KATNAL1 act co-operatively in mammalian meiosis and spermiogenesis to achieve male fertility“.
- Jess spoke at the Australian Reproduction Update meeting on “Fundamental mechanisms that drive mammalian sperm production”.
- Jess spoke at the Asia Pacific Initiative on Reproduction conference on “Molecular mechanisms of sperm production – the role of microtubules”.
- Brendan and Jess co-won the Male Contraceptive Initiative award at the annual Society for Reproductive Biology meeting.
- Jess spoke on the podcast about. Watch it here.
- Moira and Jess secured an ARC Discovery Project with collaborator Prof. Carsten Janke on “Decoding microtubule remodelling in sperm production”.
- We said goodbye to our long time research assistant Jo Merriner, who is now again working at Monash University.
- Grace Allthorpe undertook Honours with us and Gulizar Saritas is visiting from Denmark for a 6 month research project.
2022
- Moira presented the Dean’s Lecture on “Why we need to talk about male fertility!” In addition to Moira, panel members included Sarah Robertson, David Gardner and Deidre Mattiske
- Moira was featured on the Repro Radio podcast on “Sperm production and male fertility” Listen here
- Brendan spoke at the Australian Reproduction Update on the genetic causes of male fertility
- Prof. Sarah Robertson is on sabbatical with our lab here in Melbourne. It is wonderful to have Sarah around.
- A very important study was co-published with collaborators Joris Veltman, Manon Oud and team: “A de novo paradigm for male infertility“
- We published our article Human INHBB gene variant (c. 1079T> C: p. Met360Thr) alters testis germ cell content, but does not impact fertility in mice in Endocrinology with collaborators including Kelly Walton
- Maddison Graffeo started her PhD and Joanna Jiang undertook Honours with us.
- Brendan was elected the Society for Reproductive Biology ECR representative 2023-2024.
2021
- Joseph Nguyen started as a research assistant in the lab and brings a strong background in fly husbandry and molecular techniques.
- Our review on the validated genetic causes of human male infertility has been published by Human Reproduction Update. See more here. The project was a collaboration of the IMIGC.
- Abstracts by Gemma and Amy have been accepted for award sessions (David Healy and ART Lab Solutions) at the annual Society for Reproductive Biology conference.
- Sam, Brendan and Jess are all presenting oral presentations in the ‘Sperm Biology’ session at the SRB conference.
- We published a review on delta and epsilon tubulin in mammalian development in Trends in Cell Biology. See more here.
- Gemma’s art made it to the cover of Trends in Cell Biology